Physics, 25.12.2019 01:31, genyjoannerubiera
`suppose you have a helium balloon that is holding 2.00 l of he at a pressure of 15.18 psi. if it's temperature started out as 310.15 k, but was heated to 331.15 k, resulting in an expansion of volume to 3.24 l, what is the final pressure in atm?

Answers: 1
Other questions on the subject: Physics

Physics, 22.06.2019 10:30, legendman27
You are given two vectors a⃗ =−3.00ι^ 5.00j^ and b⃗ =5.00ι^ 2.00j^. let the counterclockwise angles be positive.
Answers: 3

Physics, 22.06.2019 10:50, afonseca73
Asheet of steel 1.5 mm thick has nitrogen atmospheres on both sides at 1200oc and is permitted to achieve a steady-state diffusion condition. the diffusion coefficient for nitrogen in steel at this temperature is 6 x 10- 11 m2 /s, and the diffusion flux is found to be 1.2 x 10-7 kg/m2 -s. also, it is known that the concentration of nitrogen in the steel at the high-pressure surface is 4 kg/m3 . how far into the sheet from this high-pressure side will the concentration be 2.0 kg/m3 ? assume a linear concentration profile.
Answers: 3


Physics, 22.06.2019 16:40, scully1442
Acapacitor is storing energy of 3 joules with a voltage of 50 volts across its terminals. a second identical capacitor of the same value is storing energy of 1 joule. what is the voltage across the terminals of the second capacitor?
Answers: 3
Do you know the correct answer?
`suppose you have a helium balloon that is holding 2.00 l of he at a pressure of 15.18 psi. if it's...
Questions in other subjects:


Social Studies, 05.07.2019 05:30

Mathematics, 05.07.2019 05:30


Mathematics, 05.07.2019 05:30



History, 05.07.2019 05:30


Mathematics, 05.07.2019 05:30


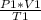 =
=