Physics, 20.12.2019 19:31, unicornpoop54
An 80.0-g piece of copper, initially at 295°c, is dropped into 250 g of water contained in a 300-g aluminum calorimeter; the water and calorimeter are initially at 10.0°c.
what is the final temperature of the system? (specific heats of copper and aluminum are 0.092 0 and 0.215 cal/g⋅°c, respectively. cw = 1.00 cal/g°c)
a. 12.8°c
b. 16.5°c
c. 28.4°c
d. 32.1°c

Answers: 2
Other questions on the subject: Physics


Physics, 22.06.2019 18:00, sarahidan
Aprisoner is forced to go into one of three rooms, but he can choose which room. the first room is ablaze with fire. the second one is rigged with explosives that will go off as soon as he enters. the third contains a pair of lions who haven't eaten in years. which room should he choose to survive?
Answers: 2

Physics, 22.06.2019 18:30, Jasten
Arailroad car collides with and sticks to an identical railroad car that is initially at rest. after the collision, the total kinetic energy of the two cars is a) the same as before. b) half as much as before. c) one third as much as before. d) one fourth as much as before. e) twice as much as before.
Answers: 1
Do you know the correct answer?
An 80.0-g piece of copper, initially at 295°c, is dropped into 250 g of water contained in a 300-g a...
Questions in other subjects:


Biology, 03.07.2020 17:01


Social Studies, 03.07.2020 17:01


Mathematics, 03.07.2020 17:01





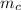 = mass of piece of copper = 80 g
= mass of piece of copper = 80 g 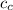 = specific heat of piece of copper = 0.0920 cal/g°C
= specific heat of piece of copper = 0.0920 cal/g°C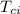 = Initial temperature of piece of copper = 295 °C
= Initial temperature of piece of copper = 295 °C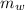 = mass of water = 250 g
= mass of water = 250 g 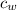 = specific heat of water = 1 cal/g°C
= specific heat of water = 1 cal/g°C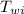 = Initial temperature of piece of copper = 10 °C
= Initial temperature of piece of copper = 10 °C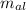 = mass of calorimeter = 300
= mass of calorimeter = 300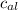 = specific heat of calorimeter = 0.215 cal/g°C
= specific heat of calorimeter = 0.215 cal/g°C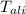 = Initial temperature of calorimeter = 10 °C
= Initial temperature of calorimeter = 10 °C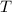 = Final equilibrium temperature
= Final equilibrium temperature