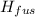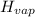Physics, 17.12.2019 01:31, Paulalex8765
How much heat energy is required to vaporize a 1.0-g ice cube at 0°c? the heat of fusion of ice is 80 cal/g.
the heat of vaporization of water is 540 cal/g, and cwater = 1.00 cal/g⋅°c.
a. 620 cal
b. 720 cal
c. 820 cal
d. 1 kcal

Answers: 3
Other questions on the subject: Physics

Physics, 22.06.2019 03:50, ash011519
The force acting on a beam was measured under the same operating conditions and a sample of 5 data points was collected. this process was repeated by 3 observers. the pooled standard deviations of these 3 data sets was 1.21. determine the true mean force using all pooled data (with a 95% probability). the result should look like this: true mean = mean +/- margin of error
Answers: 1

Physics, 22.06.2019 10:30, austinwst3
You are driving directly behind a pickup truck, going at the same speed as the truck. a crate falls from the bed of the truck to the road. (a) will your car hit the crate before the crate hits the road if you neither brake nor swerve? (b) during the fall, is the horizontal speed of the crate more than, less than, or the same as that of the truck?
Answers: 2

Do you know the correct answer?
How much heat energy is required to vaporize a 1.0-g ice cube at 0°c? the heat of fusion of ice is...
Questions in other subjects:


Business, 27.08.2020 09:01

Medicine, 27.08.2020 09:01

Mathematics, 27.08.2020 09:01

Mathematics, 27.08.2020 09:01


Mathematics, 27.08.2020 09:01

Biology, 27.08.2020 09:01

Engineering, 27.08.2020 09:01