
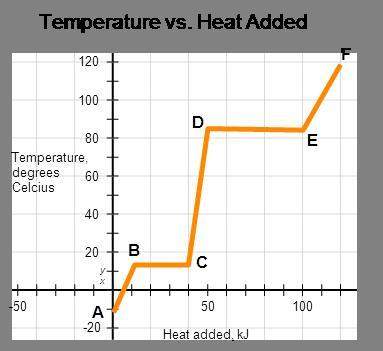
The represented substance has the following heat capacities, enthalpy of fusion, and enthalpy of vaporization.
cp, solid = 3.2 j/(g•°c); cp, liquid = 5.3 j/(g•°c);
cp, vapor = 8.9 j/(g•°c); δhfus = 4.5 kj/mol;
δhvap = 8.6 kj/mol
calculate the total energy input required to accomplish vaporization of one mole of substance at 15°c. the molar mass of the substance is 120.0 g/mol.

Answers: 2
Other questions on the subject: Physics

Physics, 21.06.2019 19:30, yolinda123429
Molten iron fills a mould, which has a volume of 200 cm cubed. calculate the volume when the iron cools and solidifies.
Answers: 1


Physics, 22.06.2019 09:30, abdominguez7187
An electric clothes dryer has a resistance of 8 ohms. it draws 30 a of current. what is the voltage, in volts, of the wall outlet that it is plugged into?
Answers: 2

Physics, 22.06.2019 10:20, studyowl9192
Electromagnetic induction. a coil of wire contains n turns and has an electrical resistance r. the radius of each turn is a. initially, inside the coil there exists a uniform magnetic field of magnitude b0 parallel to the axis of the coil. the magnetic field is then reduced slowly. the current induced in the coil is i. how long does it take for the magnitude of the uniform field to drop to zero?
Answers: 1
Do you know the correct answer?
The represented substance has the following heat capacities, enthalpy of fusion, and enthalpy of vap...
Questions in other subjects:




Mathematics, 15.10.2019 02:10

English, 15.10.2019 02:10

Chemistry, 15.10.2019 02:10

Mathematics, 15.10.2019 02:10


English, 15.10.2019 02:10






