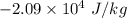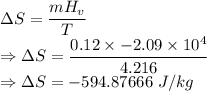Physics, 09.12.2019 18:31, starfox5454
What is the change in entropy of helium gas with total mass 0.120 kg at the normal boiling point of helium when it all condenses isothermally to liquid helium? assume that the normal boiling point of helium is 4.216 k and the heat of vaporization of helium is 2.09×10^4 j/kg .

Answers: 2
Other questions on the subject: Physics

Physics, 22.06.2019 03:50, am2garcia5
A30 kg weight lies on top of a massless piston of area a = 0.01 m2 the exterior air is at a (constant) p =1 atm and t = 27 c. the interior gas is 0.4 moles of (ideal) n2 and it has initial temperature 27.00 degrees c. 1. what is the initial pressure in the interior? a. 29.4 kpa b. 130.7 kpa c. 101.3 kpa the next three questions concern what happens when an amount of heat q is slowly added to the interior, raising the piston by 1 mm and raising the interior temperature to 27.40 c
Answers: 3

Physics, 22.06.2019 08:30, 2021arabellacorsino
An object weigh 40n in air ,weigh 20n when submerged in water, and 30n when submerged in a liquid of unknown liquid density. what is the density of unknown of liquid?
Answers: 1

Physics, 22.06.2019 10:00, myanniespencer39
Which accurately compares concave and convex lenses?
Answers: 2
Do you know the correct answer?
What is the change in entropy of helium gas with total mass 0.120 kg at the normal boiling point of...
Questions in other subjects:

Mathematics, 20.02.2020 07:57


Physics, 20.02.2020 07:58

Health, 20.02.2020 07:58

Health, 20.02.2020 07:58






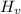 = Heat of vaporization =
= Heat of vaporization =