
Physics, 05.12.2019 03:31, lexhorton2002
The combined statement of the first and second laws for the change in enthalpy of unary single-phase system may be written: dh' = tds' +v'dp +udn use this result to write an expression for the change in enthalpy of a two-phase (alpha + beta) system. if the entropy, pressure, and total number of moles are constrained to be constant, then the criterion for equilibrium is that the enthalpy is a minimum. paraphrase the strategy used to deduce the conditions for equilibrium in an isolated system to derive them for a system constrained to constant s', p and n. what happens to the condition for mechanical equilibrium?

Answers: 2
Other questions on the subject: Physics



Physics, 22.06.2019 07:30, carlinryan
Which of the following is an example of motion in two dimensions?
Answers: 3
Do you know the correct answer?
The combined statement of the first and second laws for the change in enthalpy of unary single-phase...
Questions in other subjects:

Health, 13.12.2020 09:00


Mathematics, 13.12.2020 09:00



Spanish, 13.12.2020 09:00





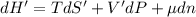
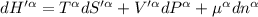
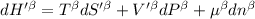
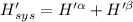
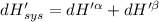
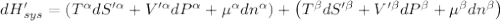
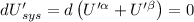
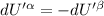
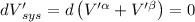
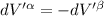
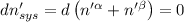
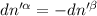
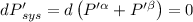
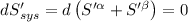
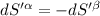
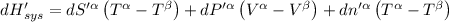
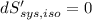
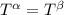 ----- for thermal equilibrium
----- for thermal equilibrium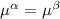 ----- for chemical equilibrium
----- for chemical equilibrium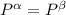 ----- for mechanical equilibrium
----- for mechanical equilibrium 



