
In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used to determine the specific heat of a solid, or to measure the energy of a solution phase reaction. a student heats 69.12 grams of tungsten to 98.93 °c and then drops it into a cup containing 85.45 grams of water at 23.82 °c. she measures the final temperature to be 25.63 °c. the heat capacity of the calorimeter (sometimes referred to as the calorimeter constant) was determined in a separate experiment to be 1.56 j/°c. assuming that no heat is lost to the surroundings calculate the specific heat of tungsten.

Answers: 1
Other questions on the subject: Physics

Physics, 22.06.2019 07:00, emmalado45
The table below shows the acceleration of gravity on different bodies in the solar system. on which body would a 10 kg lamp has the most gravitational potential energy when lifted to a height of 2 m? a. earth b. mars c. neptune d. uranus
Answers: 1

Physics, 22.06.2019 17:30, madisonnewby5021
Four point charges each having charge q are located at the corners of a square having sides of length a. find symbolic expressions for the following. (a) the total electric potential at the center of the square due to the four charges (use any variable or symbol stated above along with the following as necessary: ke.) vtotal= (b) the work required to bring a fifth charge p from infinity to the center of the square (use any variable or symbol stated above along with the following as necessary: w=
Answers: 2
Do you know the correct answer?
In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used t...
Questions in other subjects:




English, 22.10.2019 02:50

Social Studies, 22.10.2019 02:50



Mathematics, 22.10.2019 02:50


Mathematics, 22.10.2019 02:50


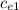 = 128.3 J / kg ° C
= 128.3 J / kg ° C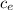 ΔT
ΔT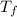 ) = (m₂
) = (m₂ 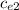 + C) (
+ C) (



