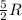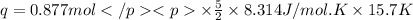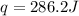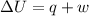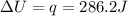A0.877 mol sample of n2(g) initially at 298 k and 1.00 atm is held at constant volume while enough heat is applied to raise the temperature of the gas by 15.7 k. assuming ideal gas behavior, calculate the amount of heat in joules required to affect this temperature change and the total change in internal energy, δ note that some books use δ as the symbol for internal energy instead of δ

Answers: 3
Other questions on the subject: Physics

Physics, 22.06.2019 12:00, jasondesatnick
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 3

Do you know the correct answer?
A0.877 mol sample of n2(g) initially at 298 k and 1.00 atm is held at constant volume while enough h...
Questions in other subjects:

Mathematics, 11.09.2020 01:01

English, 11.09.2020 01:01

Social Studies, 11.09.2020 01:01

Mathematics, 11.09.2020 01:01

Mathematics, 11.09.2020 01:01

Mathematics, 11.09.2020 01:01

Social Studies, 11.09.2020 01:01

Social Studies, 11.09.2020 01:01

Mathematics, 11.09.2020 01:01

English, 11.09.2020 01:01


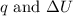 is 286.2 J and 286.2 J respectively.
is 286.2 J and 286.2 J respectively.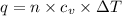
 = Change in temperature = 15.7 K
= Change in temperature = 15.7 K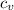 = heat capacity at constant volume of
= heat capacity at constant volume of 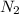 (diatomic molecule) =
(diatomic molecule) =