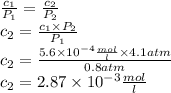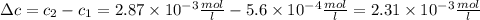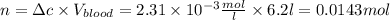The solubility of n2 in blood at 37°c and a partial pressure of 0.80 atm is 5.6 ✕ 10−4 mol·l−1. a deep-sea diver breathes compressed air with a partial pressure of n2 equal to 4.1 atm. assume that the total volume of blood in this diver's body is 6.2 l. calculate the amount of n2 gas released (in liters) when the diver returns to the surface of water, where the partial pressure of n2 is 0.80 atm. (2 sig fig)

Answers: 3
Other questions on the subject: Physics

Physics, 22.06.2019 08:30, lovebunfan
What is the primary force that suspension bridges use cables to hold their spans up? a. tension force b. resistance force c. normal force d. elastic force e. applied force
Answers: 3


Physics, 22.06.2019 19:30, cakey3501
The nuclear potential that binds protons and neutrons in the nucleus of an atom is often approximated by a square well. imagine a proton conned in an innite square well of length 105 nm, a typical nuclear diameter. calculate the wavelength and energy associated with the photon that is emitted when the proton undergoes a transition from the rst excited state (n 2) to the ground state (n 1). in what region of the electromagnetic spectrum does this wavelength belong?
Answers: 1

Physics, 23.06.2019 00:30, bradydodson47
Language is not the only way that humans communicate. describe how we use sound, touch, chemicals, and sight to communicate. need answer asap
Answers: 2
Do you know the correct answer?
The solubility of n2 in blood at 37°c and a partial pressure of 0.80 atm is 5.6 ✕ 10−4 mol·l−1. a de...
Questions in other subjects:


Mathematics, 06.11.2019 02:31


Mathematics, 06.11.2019 02:31

Biology, 06.11.2019 02:31


Mathematics, 06.11.2019 02:31