
Physics, 01.11.2019 01:31, angeldawnfick
Abackpacker collects snow at 0°c, and places it in a cooking pot on a camp stove. it takes 643 kj of heat energy to melt the snow and bring the water to boiling. assuming no heat loss, and neglecting the specific heat capacity of the pot, calculate the mass of snow that the backpacker collected. (the specific heat capacity of liquid water, c = 4.18 j/g. k; and: h₂o(s) -> h₂o(l) /\h = /\hfusion = 6.02 kj/mol)a) 1.92 kg b) 1.90 kg c) 1.52 kg d) 855g e) < 800g

Answers: 2
Other questions on the subject: Physics

Physics, 21.06.2019 23:00, hunterdylan01
Acoal-fired plant generates 600 mw of electric power. the plant uses 4.8 x 10^6 kg of coal each day, and the heat of combustion of coal is 3.3x 10^7 j/kg. the steam that drives the turbines is at a temperature of 300° c, and the exhaust water is at 37° c. (a) what is the overall efficiency of the plant for generating electric power? (b) how much thermal energy is exhausted each day? (c) what is the change in entropy of the universe for this heat engine? (d) using the same heat reservoirs, what is the maximum possible (carnot) efficiency for a heat engine?
Answers: 1



Physics, 22.06.2019 22:00, savannahworkman11
Acar is traveling 35 mph on a smooth surface . if a balanced force is applied to the car what happens?
Answers: 3
Do you know the correct answer?
Abackpacker collects snow at 0°c, and places it in a cooking pot on a camp stove. it takes 643 kj of...
Questions in other subjects:











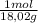 ×mass
×mass




