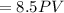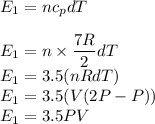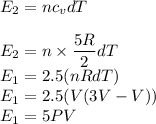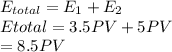Asample of a diatomic ideal gas has pressure p and volume v. when the gas is warmed, its pressure triples and its volume doubles. this warming process includes two steps, the first at constant pressure and the second at constant volume. determine the amount of energy transferred to the gas by heat. (here we define a "diatomic ideal gas" to havecv =52randcp =72r. use any variable or symbol stated above as necessary.)

Answers: 1
Other questions on the subject: Physics

Physics, 22.06.2019 02:30, jendun123ovrxij
The particle in a two-dimensional well is a useful model for the motion of electrons around the indole ring (3), the conjugated cycle found in the side chain of tryptophan. we may regard indole as a rectangle with sides of length 280 pm and 450 pm, with 10 electrons in the conjugated p system. as in case study 9.1, we assume that in the ground state of the molecule each quantized level is occupied by two electrons. (a) calculate the energy of an electron in the highest occupied level. (b) calculate the frequency of radiation that can induce a transition between the highest occupied and lowest unoccupied levels. 9.27 electrons around the porphine ring (4), the conjugated macrocycle that forms the structural basis of the heme group and the chlorophylls. we may treat the group as a circular ring of radius 440 pm, with 20 electrons in the conjugated system moving along the perimeter of the ring. as in exercise 9.26, assume that in the ground state of the molecule quantized each level is occupied by two electrons. (a) calculate the energy and angular momentum of an electron in the highest occupied level. (b) calculate the frequency of radiation that can induce a transition between the highest occupied and lowest unoccupied levels.
Answers: 1



Do you know the correct answer?
Asample of a diatomic ideal gas has pressure p and volume v. when the gas is warmed, its pressure tr...
Questions in other subjects:




Mathematics, 15.01.2021 20:40




Mathematics, 15.01.2021 20:40


Mathematics, 15.01.2021 20:40