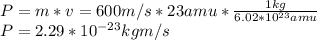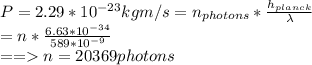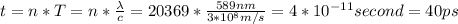Physics, 14.09.2019 08:30, emilylunaa
Asodium atom will absorb light with a wavelength near 589 nm if the light is within 10 mhz of the resonant frequency. the atomic mass of sodium is 23. (i) calculate the number of "yellow" photons of wavelength 2 = 589 nm that must be absorbed to stop a sodium atom initially at room temperature (v-600 m/s). [7 marks] (ii) what is the minimum time needed to cool a sodium atom?

Answers: 3
Other questions on the subject: Physics


Physics, 22.06.2019 00:20, glogaming16
In the derivation of rrkm theory, a factor of 1/2 is introduced when equalizing the rates of formation and decomposition of activated complex as keal-hr) = ko this is clearly against the assumption of transition state theory that states all the activated complex in the transition state iss going to the product. find the reason why this factor is introduced here.
Answers: 2

Physics, 22.06.2019 14:40, akitchen10
A5 foot by 5 foot box culvert is buried 6 feet beneath the surface of the ground. a crosssection of this very long culvert (into and out of the page) is illustrated below. assume that the soil in which the culvert is buried has the following properties: jt = 120 pcf, w = 12%, ko = 0.50, ka = 0.33, and kp = 3.00. calculate the total horizontal force per unit length of culvert (fh) felt on one of the vertical faces of the culvert.
Answers: 1
Do you know the correct answer?
Asodium atom will absorb light with a wavelength near 589 nm if the light is within 10 mhz of the re...
Questions in other subjects:


Chemistry, 13.03.2021 09:20

Engineering, 13.03.2021 09:20



Mathematics, 13.03.2021 09:20

Mathematics, 13.03.2021 09:20


Mathematics, 13.03.2021 09:20

English, 13.03.2021 09:20