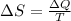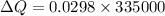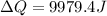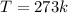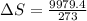Physics, 12.09.2019 22:30, Alexis3456829
What is the entropy change of a 29.8 g ice cube that melts completely in a bucket of water whose temperature is just above the freezing point of water?

Answers: 1
Other questions on the subject: Physics



Physics, 22.06.2019 14:50, MoogleCaliS
Nitrogen (n2) undergoes an internally reversible process from 6 bar, 247°c during which pν1.2 = constant. the initial volume is 0.1 m3 and the work for the process is 121.14 kj. assuming ideal gas behavior, and neglecting kinetic and potential energy effects, determine heat transfer, in kj, and the entropy change, in kj/s. show the process on a t-s diagram.
Answers: 2
Do you know the correct answer?
What is the entropy change of a 29.8 g ice cube that melts completely in a bucket of water whose tem...
Questions in other subjects:






Mathematics, 06.11.2019 04:31



Biology, 06.11.2019 04:31

Biology, 06.11.2019 04:31