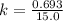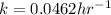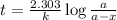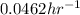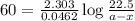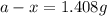Answers: 1
Other questions on the subject: Physics

Physics, 22.06.2019 08:00, hartzpeyton136
5g of ammonium nitrate was dissolved in 60g of water in an insulated container. the temperature at the start of the reaction was 23.0°c and at the end it was 19.0°c. calculate the energy absorbed by the reaction.
Answers: 3

Physics, 22.06.2019 16:30, sti192
The air in an automobile tire with a volume of 2.60 ft3 is at 70°f and 21 psig. determine the amount of air that must be added to raise the pressure to the recommended value of 30 psig. assume the atmospheric pressure to be 14.6 psia and the temperature and the volume to remain constant. the gas constant of air is ru = 53.34ft⋅lbflbm⋅r(1 psia144 lbf/ft2) = 0.3704 psia⋅ft3lbm⋅r the amount of air that must be added to raise the pressure is lbm
Answers: 3

Physics, 23.06.2019 10:30, tamikagoss22
Can someone answer my last question i posted? on my last post? my hw is due tomorrow and i’m confused.
Answers: 1
Do you know the correct answer?
Sodium-24, which is used to locate blood clots in the human circulatory system, has a half-life of 1...
Questions in other subjects:

Mathematics, 30.10.2020 04:20



Mathematics, 30.10.2020 04:20




English, 30.10.2020 04:30

English, 30.10.2020 04:30