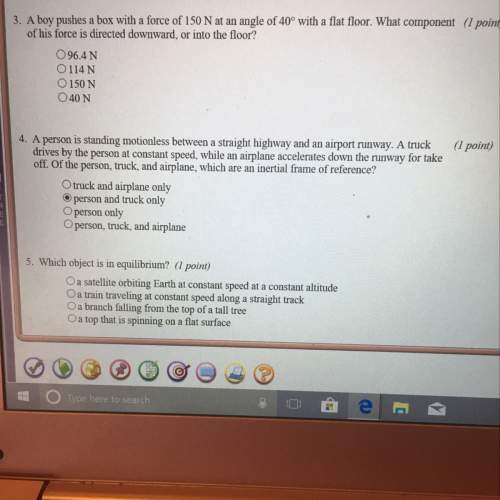
Answers: 3
Other questions on the subject: Physics


Physics, 22.06.2019 10:00, lorelei7668
(a) calculate the number of electrons in a small, electrically neutral silver pin that has a mass of 10.0 g. silver has 47 electrons per atom, and its molar mass is 107.87 g/mol. (b) imagine adding electrons to the pin until the negative charge has the very large value 1.00 mc. how many electrons are added for every 109 electrons already present
Answers: 3

Physics, 22.06.2019 12:30, morgan3346
Write a full page that sumerizes thermodynamics it’s from the website visionlearnig
Answers: 1

Physics, 22.06.2019 12:50, angelica3752
Match each vocabulary term to its definition. 1. electrons neutral subatomic particles found in the nucleus of the atom 2. neutron lowest energy position of an electron in an atom 3. photon the path of an electron around the nucleus of an atom 4. ground state negatively charged, subatomic particles 5. protons packet of energy of specific size 6. element substance with only one type of atom 7. orbital positively charged, subatomic particles found in the nucleus of the atom
Answers: 1
Do you know the correct answer?
Describe the evolution of a pulsar over time, in particular how the rotation and pulse signal change...
Questions in other subjects:


Social Studies, 07.10.2019 21:00



History, 07.10.2019 21:00

Social Studies, 07.10.2019 21:00


Biology, 07.10.2019 21:00

Mathematics, 07.10.2019 21:00