Physics, 05.08.2019 16:10, dejaproctor17
To find the specific heat capacity of a certain metal, a student places a block of the metal that weighs 27 grams and has an initial temperature of 97 deg c, into 52 grams of water with a temperature of 20 deg c. the final temperature was measured to be 27 deg c. what is the heat capacity of the metal?

Answers: 1
Other questions on the subject: Physics

Physics, 21.06.2019 13:30, 214578
An air bubble of volume 20 cm³ is at the bottom of a lake 40 m deep, where the temperature is 4.0°c. the bubble rises to the surface, which is at a temperature of 20°c. take the temperature of the bubble’s air to be the same as that of the surrounding water. just as the bubble reaches the surface, what is its volume?
Answers: 1

Physics, 22.06.2019 21:00, tobyfoerst
If the specific heat of a metal is 0.850 j/g °c, what is its atomic weight?
Answers: 2


Physics, 23.06.2019 02:00, hgg44137
Employer informs its employees that it will hold a substantial cash prize drawing for each work group at the end of each month in which no employee in the work group sustains a lost-time injury. tyra reports an injury that she sustained while operating a mechanical power press. tyra did not violate any employer safety rules when she sustained her injury. tyra’s injury requires her to miss work for two days. her employer cancels the cash prize drawing for that month for tyra’s work group because of tyra’s lost-time injury. did the employer misuse an incentive program to retaliate against tyra?
Answers: 3
Do you know the correct answer?
To find the specific heat capacity of a certain metal, a student places a block of the metal that we...
Questions in other subjects:

Mathematics, 27.05.2021 21:40

Computers and Technology, 27.05.2021 21:40



Mathematics, 27.05.2021 21:40





Mathematics, 27.05.2021 21:40


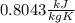
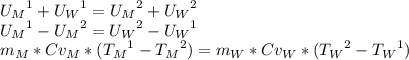
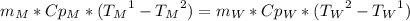
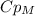 , so:
, so: