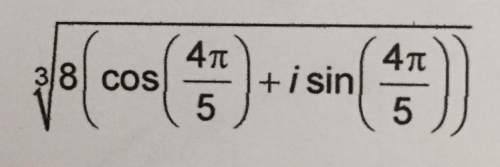Mathematics, 09.04.2021 17:40, 5001Je
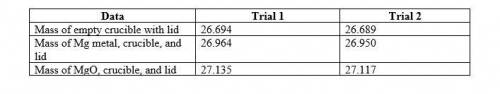
1. Write the balanced chemical equation for the reaction you are performing. 4. Magnesium is the limiting reactant in this experiment. Calculate the theoretical yield of MgO for each trial.
• Trial 1:
• Trial 2:
5. Determine the percent yield of MgO for your experiment for each trial.
• Trial 1:
• Trial 2:
6. Determine the average percent yield of MgO for the two trials.

Answers: 2
Other questions on the subject: Mathematics


Mathematics, 21.06.2019 19:10, happy121906
With this question substitute t=3 and t=5 to determine if the two expressions are equivalent. 4(t + 3) 4t+12 which statements are true? check all that apply. a. the value of both expressions when t =5 is 32. b. the two expressions are not equivalent. c. the value of both expressions when t=3 is 15. d. the value of both expressions when t=5 is 23. e. the two expressions are equivalent. f. the value of both expressions when t=3 is 24.
Answers: 3


Mathematics, 22.06.2019 01:30, Queenempress9380
A23 – year – old male buys 100/300/100 liability insurance, collision insurance with a $100 deductible, and comprehensive insurance with a $250 deductible? what is his total annual premium? (a) $1142.00 (b) $1765.45 (c) $1770.10 (d) $1785.25
Answers: 2
Do you know the correct answer?
1. Write the balanced chemical equation for the reaction you are performing. 4. Magnesium is the lim...
Questions in other subjects:





Mathematics, 25.02.2021 17:50



English, 25.02.2021 17:50