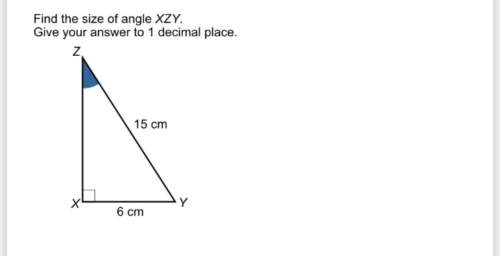
Mathematics, 04.12.2020 17:00, emiller6462
The wave function of an electron in the lowest (that is, ground) state of the hydrogen atom is
φ (r)= (1/ πao^3) exp (-r/ao)
Required:
a. What is the probability of finding the electron inside a sphere of volume 1.0 pm^3 , centered at the nucleus.
1pm= 10^-12m
b. What is the probability of finding the electron in a volume of 1.0 pm3 at a distance of 52.9 pm from the nucleus, in a fixed but arbitrary direction?
c. What is the probability of finding the electron in a spherical shell of 1.0 pm in thickness, at a distance of 52.9 pm from the nucleus?

Answers: 1
Other questions on the subject: Mathematics

Mathematics, 21.06.2019 17:00, maddiehope6140
Acar travels at an average speed of 56 miles per hour. how long does it take to travel 196 miles
Answers: 1


Do you know the correct answer?
The wave function of an electron in the lowest (that is, ground) state of the hydrogen atom is
φ (r...
Questions in other subjects: