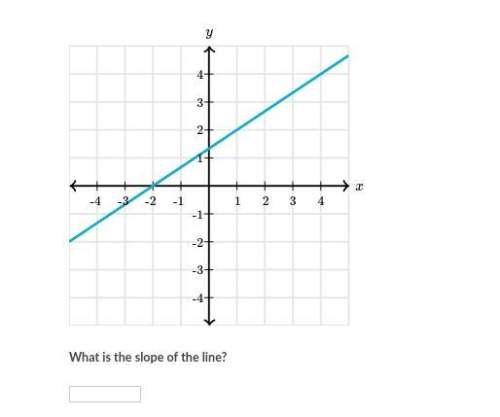
Mathematics, 01.04.2020 21:39, BIGJAYWESTSIDE
At a temperature of 10°C, 700 ml of hydrogen is collected. If this gas is put into a 1,000-ml container, what will it’s new temperature be in Celsius?
At 1,000°C and a temperature of 1,200 mm Hg, a sample of methane occupies 20.0 l. What will be it’s new temperature if it is compressed to a volume of 4.00 l and a pressure of 3,600 mm Hg.
A sample of nitrous oxides has a volume of 120 l at 1,500 mm Hg and -10°C. Calculate its pressure when it’s volume is compressed to 50 l and it’s temperature increased to 34°C.
At 27°C and 750 mm Hg, a sample of hydrogen occupies 5.00 l. How much space will it occupy at STP?
The density of hydrogen at STP is 0.09 g/l. What is the mass of the sample of hydrogen in the last question.

Answers: 2
Other questions on the subject: Mathematics

Mathematics, 21.06.2019 17:00, nisazaheer
Determine the number of outcomes in the event. decide whether the event is a simple event or not. upper a computer is used to select randomly a number between 1 and 9 comma inclusive. event upper b is selecting a number greater than 4. event upper b has nothing outcome(s). is the event a simple event? (yes or no) because event upper b has (fewer than, exactly, more than) one outcome.
Answers: 1


Do you know the correct answer?
At a temperature of 10°C, 700 ml of hydrogen is collected. If this gas is put into a 1,000-ml contai...
Questions in other subjects:

Spanish, 17.07.2020 23:01




Mathematics, 17.07.2020 23:01