
Engineering, 18.02.2021 07:10, mrus
A petrochemical plant in Island C produces formaldehyde (CH2O) industrially by the
catalytic oxidation of methanol (CH3OH). The following is the chemical reaction:
CH3OH + ½O2 → CH2O + H2O --- (1)
Unfortunately, a significant portion of the formaldehyde will react with oxygen to produce CO
and H2O. The following is the side reaction:
CH2O + ½O2 → CO + H2O --- (2)
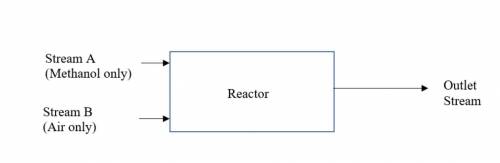
Assume twice the stoichiometric amount of air needed for oxidation (all chemical reactions) is
fed to the reactor. The conversion of methanol is 90% in the reactor. The outlet stream of the
reactor was analysed and found to be 150 kg/hr of formaldehyde, 30 kg/hr of CO and other
components.(i) Determine the mass flowrate (kg/hr) of Stream A. (ii) Determine the molar flowrate (kg-moles/hr) of Stream B (iii) Determine the mass flowrate (kg/hr) of water in the outlet stream.

Answers: 3
Other questions on the subject: Engineering

Engineering, 04.07.2019 18:10, agpraga23ovv65c
Carbon dioxide gas expands isotherm a turbine from 1 mpa, 500 k at 200 kpa. assuming the ideal gas model and neglecting the kinetic and potential energies, determine the change in entropy, heat transfer and work for each kilogram of co2.
Answers: 2

Engineering, 04.07.2019 18:10, Strick1530
Which of the following components of a pid controlled accumulates the error over time and responds to system error after the error has been accumulated? a)- proportional b)- derivative c)- integral d)- on/off.
Answers: 2


Engineering, 04.07.2019 19:10, nbunny7208
What is the chief metrological difference between measuring with a microscope and with an electronic comparator? a. the microscope is limited to small workpieces. a. the microscope is limited to small workpieces. c. the comparator can only examine one point on the workpiece. d. the microscope carries its own standard.
Answers: 1
Do you know the correct answer?
A petrochemical plant in Island C produces formaldehyde (CH2O) industrially by the
catalytic oxidat...
Questions in other subjects:


Mathematics, 27.02.2020 01:53


Mathematics, 27.02.2020 01:53



Mathematics, 27.02.2020 01:53

English, 27.02.2020 01:53

Biology, 27.02.2020 01:53







