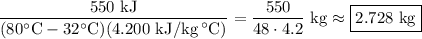Engineering, 06.11.2020 23:40, lakeithiat1320
A 550 kJ of heat quantity needed to increase water temperature from 32°C to 80°C. Calculate the mass of the water when the specific heat capacity of water is 4200 J/kg °C.

Answers: 1
Other questions on the subject: Engineering

Engineering, 03.07.2019 14:10, makaylashrout77
Amass of 1.5 kg of air at 120 kpa and 24°c is contained in a gas-tight, frictionless piston-cylinder device. the air is now compressed to a final pressure of 720 kpa. during the process, heat is transferred from the air such that the temperature inside the cylinder remains constant. calculate the boundary work input during this process.
Answers: 2


Engineering, 04.07.2019 18:20, ashleyjaslin
Derive the correction factor formula for conical nozzle i=-(1+ cosa) and calculate the nozzle angle correction factor for a nozzle whose divergence hal-fangle is 13 (hint: assume that all the mass flow originates at the apex of the cone.
Answers: 3

Engineering, 04.07.2019 18:20, samantha636
Avolume of 2.65 m3 of air in a rigid, insulated container fitted with a paddle wheel is initially at 264 k, 5.6 bar. the air receives 432 kj by work from the paddle wheel. assuming the ideal gas model with cv = 0.71 kj/kg • k, determine for the air the amount of entropy produced, in kj/k
Answers: 2
Do you know the correct answer?
A 550 kJ of heat quantity needed to increase water temperature from 32°C to 80°C. Calculate the mass...
Questions in other subjects:






Mathematics, 13.05.2021 06:50

Mathematics, 13.05.2021 06:50

Advanced Placement (AP), 13.05.2021 06:50

Mathematics, 13.05.2021 06:50

Mathematics, 13.05.2021 06:50