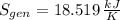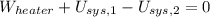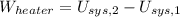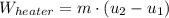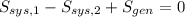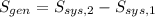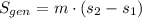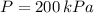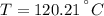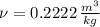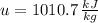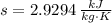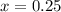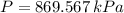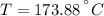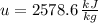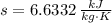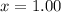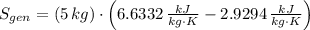Engineering, 15.04.2020 03:05, masonsee4ytube
A well-insulated rigid tank contains 5 kg of a saturated liquid–vapor mixture of water at 200 kPa. Initially, three-quarters of the mass is in the liquid phase. An electric resistance heater placed in the tank is now turned on and kept on until all the liquid in the tank is vaporized. Determine the entropy change of the steam during this process. Use steam tables.

Answers: 3
Other questions on the subject: Engineering

Engineering, 03.07.2019 14:10, aliceohern
The y form of iron is known as: a) ferrite b) cementite c) perlite d) austenite
Answers: 3

Engineering, 04.07.2019 18:10, johnthienann58
Thermal stresses are developed in a metal when its a) initial temperature is changed b) final temperature is changed c) density is changed d) thermal deformation is prevented e) expansion is prevented f) contraction is prevented
Answers: 2

Engineering, 04.07.2019 18:10, xboxdude06
Slip occurs via two partial dislocations because of (a) the shorter path of the partial dislocation lines; (b) the lower energy state through partial dislocations; (c) the charge balance.
Answers: 1

Engineering, 04.07.2019 18:20, hayleymckee
Steam enters a converging nozzle at 3.0 mpa and 500°c with a at 1.8 mpa. for a nozzle exit area of 32 cm2, determine the exit velocity, mass flow rate, and exit mach number if the nozzle: negligible velocity, and it exits (a) is isentropic (b) has an efficiency of 94 percent
Answers: 2
Do you know the correct answer?
A well-insulated rigid tank contains 5 kg of a saturated liquid–vapor mixture of water at 200 kPa. I...
Questions in other subjects:


English, 18.02.2021 05:40

History, 18.02.2021 05:40


Chemistry, 18.02.2021 05:40


History, 18.02.2021 05:40

Physics, 18.02.2021 05:40

Mathematics, 18.02.2021 05:40

Mathematics, 18.02.2021 05:40