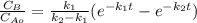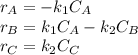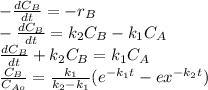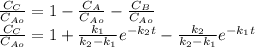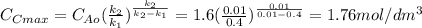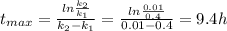The elementary liquid-phase series reaction
A k1> B k2> C
is carried out in a 500-...

Engineering, 30.03.2020 23:05, avinashpolwah
The elementary liquid-phase series reaction
A k1> B k2> C
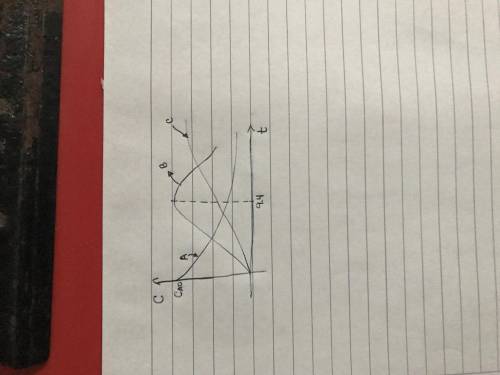
is carried out in a 500-dm^3 batch reactor. The initial concentration of A is 1.6 mol/dm^3. The desired product is B, and separation of the undesired product C is very difficult and costly. Because the reaction is carried out at a relatively high temperature, the reaction is easily quenched.
(a) Plot and analyze the concentrations of A, B, and C as a function of time. Assume that each reaction is irreversible, with k1 = 0.4 h^-1 and k2 = 0.01 h^-1.

Answers: 1
Other questions on the subject: Engineering

Engineering, 04.07.2019 18:10, caitlynnpatton1208
Water in a partially filled large tank is to be supplied to the roof top, which is 8 m above the water level in the tank, through a 2.2-cm-internal-diameter pipe by maintaining a constant air pressure of 300 kpa (gage) in the tank. if the head loss in the piping is 2 m of water, determine the discharge rate of the supply of water to the roof top in liters per second.
Answers: 3


Engineering, 04.07.2019 18:10, ayoismeisjjjjuan
Manometers are good examples of measuring instruments, nowadays they are not as common as before. a)-capacitive probe gauges b)-gravitational gauges deformation ) gauges d)-digital gauges
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Business, 12.03.2021 01:00

Arts, 12.03.2021 01:00

Arts, 12.03.2021 01:00

Chemistry, 12.03.2021 01:00

History, 12.03.2021 01:00

Mathematics, 12.03.2021 01:00

Biology, 12.03.2021 01:00

Mathematics, 12.03.2021 01:00