Engineering, 18.03.2020 22:36, salvadorjr1226p4zkp3
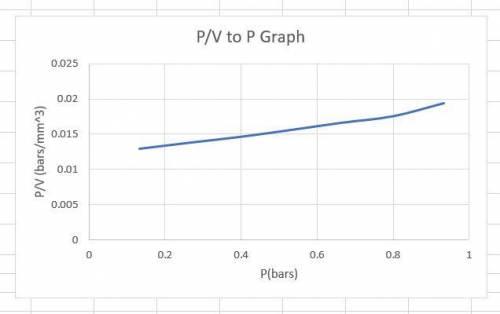
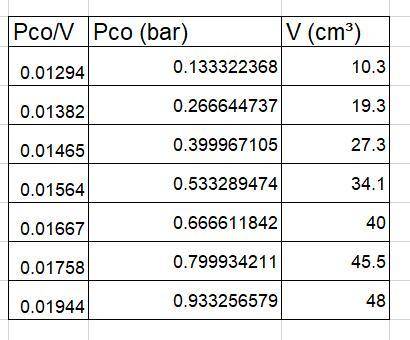
The data below were obtained for the adsorption of CO on charcoal at 273 K using an instrument that measures the volume (corrected to 1 atm pressure) of gas molecules that become adsorbed onto a surface as a function of its pressure in the surround gas phase. Such measurements are commonly performed in order to quantify the surface area of a material. The charcoal sample had a mass of 0.5 g and a CO adsorption site density of 5x10¹⁵ cm⁻².
Pco (torr) 100 200 300 400 500 600 700
V (cm³) 10.3 19.3 27.3 34.1 40 45.5 48
(a) Is the data consistent with a Langmuir isotherm? Support your answer. Hint: you will need to modify the Langmuir isotherm for a single species to convert θ_a to V.
(b) Determine the adsorption equilibrium ratio (Kco) and the volume of CO that is adsorbed at complete coverage.
(c) What is the total surface area (S_a) of the piece of charcoal in m² per gram of charcoal?

Answers: 3
Other questions on the subject: Engineering



Engineering, 04.07.2019 18:10, lowkeyqueenk
Apipe with an outside diameter of 15 cm is exposed to an ambient air and surrounding temperature of -20°c. the pipe has an outer surface temperature of 65°c and an emissivity of 0.85. if the rate of heat loss from the pipe surface is 0.95 kw per meter of length, the external convective heat transfer coefficient (h) is: (a) 12.5 w/m"k (b) 18.6 w/mk (c) 23.7 w/mk (d) 27.9 w/mk (e) 33.5 w/mk
Answers: 1

Engineering, 04.07.2019 18:10, katelynn73
Atmospheric air has a temperature (dry bulb) of 80° f and a wet bulb temperature of 60° f when the barometric pressure is 14.696 psia. determine the specific humidity, grains/lb dry air. a. 11.4 c. 55.8 d. 22.5 b. 44.1
Answers: 1
Do you know the correct answer?
The data below were obtained for the adsorption of CO on charcoal at 273 K using an instrument that...
Questions in other subjects:


English, 07.11.2020 14:00

Physics, 07.11.2020 14:00

Mathematics, 07.11.2020 14:00

Geography, 07.11.2020 14:00

Geography, 07.11.2020 14:00


History, 07.11.2020 14:00



 is 123.09 cm³
is 123.09 cm³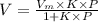
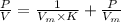 where the intercept is
where the intercept is 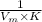 and the slope is
and the slope is 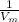
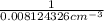 = 123.09 cm³
= 123.09 cm³ =
= 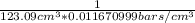 = 0.696 bar⁻¹.
= 0.696 bar⁻¹.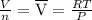 =
= 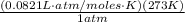 = 22.41 l
= 22.41 l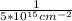 = 2 x 10⁻¹⁶ cm²
= 2 x 10⁻¹⁶ cm²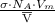 =
= 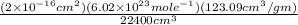 = 6.62 x 10⁵ cm²/gm
= 6.62 x 10⁵ cm²/gm