
Engineering, 13.03.2020 03:03, lolz55678
A 3.27-m3 tank contains 100 kg of nitrogen at 175 K. Determine the pressure in the tank using (a) the ideal-gas equation, (b) the van der Waals equation, and (c) the Beattie- Bridgeman equation. Compare your results with the actual value of 1505 kPa.

Answers: 3
Other questions on the subject: Engineering

Engineering, 04.07.2019 18:10, siri5645
At 12 noon, the count in a bacteria culture was 400; at 4: 00 pm the count was 1200 let p(t) denote the bacteria cou population growth law. find: (a) an expression for the bacteria count at any time t (b) the bacteria count at 10 am. (c) the time required for the bacteria count to reach 1800.
Answers: 1

Engineering, 04.07.2019 18:10, Candi9697
A-mn has a cubic structure with a0 0.8931 nm and a density of 7.47 g/cm3. b-mn has a different cubic structure, with a0 0.6326 nm and a density of 7.26 g/cm3. the atomic weight of manganese is 54.938 g/mol and the atomic radius is 0.112 nm. determine the percent volume change that would occur if a-mn transforms to b-mn.
Answers: 2


Engineering, 04.07.2019 19:10, pjgolden04
How to increase the thermal officiency of an ideal simple rankino cycle? among these methods, which one is the best and why?
Answers: 2
Do you know the correct answer?
A 3.27-m3 tank contains 100 kg of nitrogen at 175 K. Determine the pressure in the tank using (a) th...
Questions in other subjects:

Mathematics, 09.06.2021 01:00

Mathematics, 09.06.2021 01:00



English, 09.06.2021 01:00



Mathematics, 09.06.2021 01:00

Mathematics, 09.06.2021 01:00


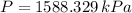 , greater than experimental value. b)
, greater than experimental value. b) 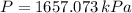 , greater than experimental value. c)
, greater than experimental value. c) 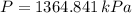 , lesser than experimental value.
, lesser than experimental value.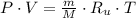
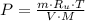
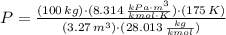
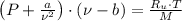 , where:
, where: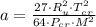 and
and 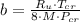
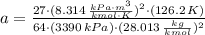
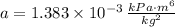
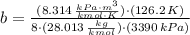
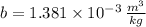
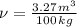
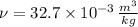
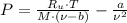
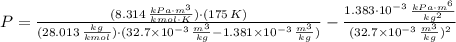
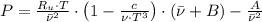
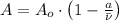 and
and 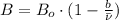 .
.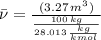
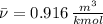
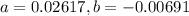
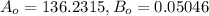
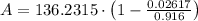
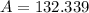
![B = 0.05046\cdot \left[ 1 - \frac{(-0.00691)}{0.916} \right]](/tpl/images/0546/0585/63324.png)
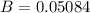
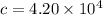
![P =\frac{(8.314\,\frac{kPa\cdot m^{3}}{kmol\cdot K} )\cdot (175\,K)}{0.916\,\frac{m^{3}}{kmol} }\cdot \left[1 - \frac{4.20\times 10^{4}}{(0.916\,\frac{m^{3}}{kmol} )\cdot (175\,K)^{3}} \right]\cdot (0.916\,\frac{m^{3}}{kmol} + 0.05084)-\frac{132.339}{(0.916\,\frac{m^{3}}{kmol} )^{2}}](/tpl/images/0546/0585/eb172.png)




