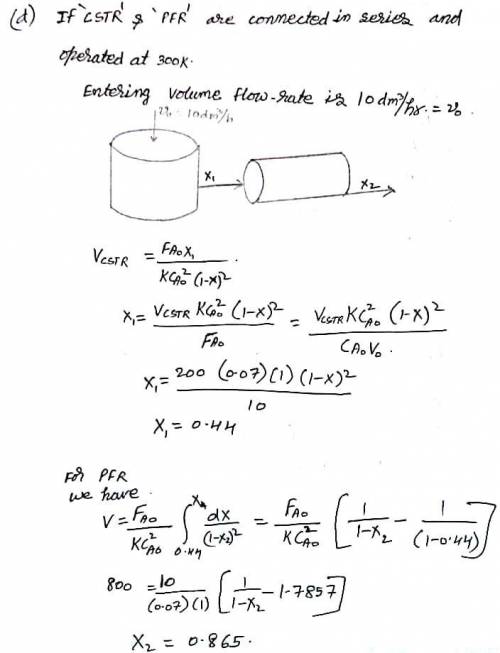
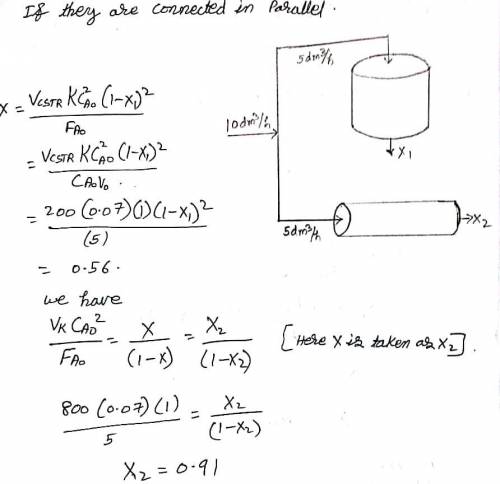
The liquid-phase reaction:
A + B → C,
follows an elementary rate law and is carried out...

Engineering, 25.02.2020 05:14, gatorlove00
The liquid-phase reaction:
A + B → C,
follows an elementary rate law and is carried out isothermally in a flow system. The concentrations of the A and B feed streams are 2 M before mixing. The volumetric flow rate of each stream is 5 dm3/min, and the entering temperature is 300 K. The streams are mixed immediately before entering. Two reactors are available. One is a gray, 200.0-dm³ CSTR that can be heated to 77C or cooled to 0C, and the other is a white, 800.0-dm³ PFR operated at 300 K that cannot be heated or cooled but can be painted red or black. Note that k 0.07 dm³ /mol-min at 300 K and E 20 kcal/mol.
Which reactor and what conditions do you recommend? Explain the reason for your choice (e. g., color, cost, space available, weather conditions). Back up your reasoning with the appropriate calculations.

Answers: 3
Other questions on the subject: Engineering

Engineering, 03.07.2019 23:20, abbz13
Two technicians are discussing the intake air temperature (iat) sensor. technician a says that the computer uses the iat sensor as a backup to the engine coolant temperature (ect) sensor. technician b says that the powertrain control module (pcm) will subtract the calculated amount of fuel if the air measures hot. who is correct
Answers: 3


Engineering, 04.07.2019 18:10, wirchakethan23
Hydraulic fluid with a sg. of 0.78 is flowing through a 1.5 in. i. d. pipe at 58 gal/min. the fluid has an absolute viscosity of 11.8 x 105 lbf-sec/ft2. is the flow laminar, turbulent or within the critical range? give both a numerical reynolds number and a term answer.
Answers: 3

Engineering, 04.07.2019 18:10, juansoto227711
Journeyman training is usually related (clo2) a)-to specific tasks b)-to cost analysis of maintenance task c)-to control process to ensure quality d)-to installation of machinery
Answers: 2
Do you know the correct answer?
Questions in other subjects: