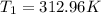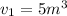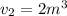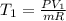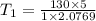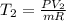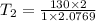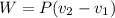Engineering, 14.02.2020 05:26, mari200150
The volume of 1 kg of helium in a piston–cylinder device is initially 5 m3. Now helium is compressed to 2 m3 while its pressure is maintained constant at 130 kPa. Determine the initial and final temperatures of helium as well as the work required to compress it, in kJ. The gas constant of helium is R = 2.0769 kJ/kg·K.

Answers: 1
Other questions on the subject: Engineering

Engineering, 04.07.2019 18:10, jadeochoa4466
The temperature of air decreases as it is compressed by an adiabatic compressor. a)- true b)- false
Answers: 2

Engineering, 04.07.2019 18:10, johnthienann58
Thermal stresses are developed in a metal when its a) initial temperature is changed b) final temperature is changed c) density is changed d) thermal deformation is prevented e) expansion is prevented f) contraction is prevented
Answers: 2

Engineering, 04.07.2019 18:10, mamasbug4285
An air compression refrigeration system is to have an air pressure of 100 psia in the brine tank and an allowable air temperature increase of 60°f for standard vapor compression cycle temperatures of 77 f entering the expansion cylinder and 14 f entering the compression cylinder, calculate the coefficient of performance a. 2.5 b 3.3 c. 4.0 d. 5.0
Answers: 3

Engineering, 04.07.2019 18:20, CelesteN64
Most leaks in reciprocating air compressors can be detected and minimized by: (clo4) a)-detecting leakage areas using ultrasonic acoustic detector. b)-tightening joints and connections c)-replacing faulty equipment d)-all of the given options
Answers: 2
Do you know the correct answer?
The volume of 1 kg of helium in a piston–cylinder device is initially 5 m3. Now helium is compressed...
Questions in other subjects:


History, 19.07.2019 08:00

Mathematics, 19.07.2019 08:00



Chemistry, 19.07.2019 08:00



Physics, 19.07.2019 08:00