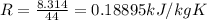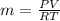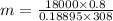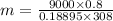Engineering, 11.10.2019 05:30, itzlianne
Carbon dioxide (co2) is used in industrial and research application, and is sometimes stored at very high pressure in rigid metal tanks. consider a tank with a volume of 0.8 m^3. (a) when the tank is first deliverd to a customer on a hot summer day, the pressure is measure to be 18,000 kpa at a temperature of 35 celsius. what the mass of co2 in the tank? (b) if the temperature remains constant, when enough co2 is used such that the tank pressure is reduced in half to 9,000 kpa, what is the mass of co2 in the tank?

Answers: 3
Other questions on the subject: Engineering

Engineering, 03.07.2019 15:10, brooklyn674
Apiston-cylinder with a volume of 0.25 m3 holds 1 kg of air (r 0.287 k/kgk) at a temperature of 100 c. heat transfer to the cylinder causes an isothermal expansion of the piston until the volume triples. how much heat is added to the piston-cylinder?
Answers: 3

Engineering, 03.07.2019 15:10, margaret1758
If you were designing a bumper for a car, would you prefer it to exhibit elastic or plastic deformation? why? consider the functions of a bumper in both a minor "fender-bender" and a major collision.
Answers: 1

Engineering, 04.07.2019 18:10, lerasteidl
Determine whether or not it is possible to compress air adiabatically from k to 140 kpa and 400 k. what is the entropy change during this process?
Answers: 3
Do you know the correct answer?
Carbon dioxide (co2) is used in industrial and research application, and is sometimes stored at very...
Questions in other subjects:

English, 13.10.2020 05:01

History, 13.10.2020 05:01



Mathematics, 13.10.2020 05:01

English, 13.10.2020 05:01


English, 13.10.2020 05:01