Chemistry, 21.07.2019 07:00, LeoInc6806
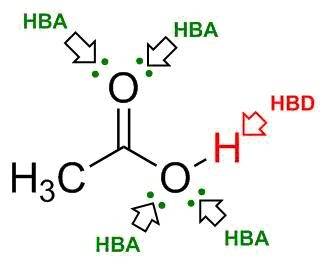
Ch3cooh (acetic acid) can form hydrogen bonds between its molecules. based on the lewis structure shown below, how many hydrogen bond donor and acceptor atoms does this molecule have?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, milkshakegrande101
Arock can be broken down into different kinds of substances by physical processes. no chemical reactions are needed to separate different parts of a rock into pure substances. this is because a rock is a(n)
Answers: 1

Chemistry, 22.06.2019 14:00, jivsf
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 16:00, winnie45
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 17:20, alexis3060
How do you know when a chemical reaction has occurred
Answers: 1
Do you know the correct answer?
Ch3cooh (acetic acid) can form hydrogen bonds between its molecules. based on the lewis structure sh...
Questions in other subjects:




Biology, 07.10.2019 18:30


English, 07.10.2019 18:30

Mathematics, 07.10.2019 18:30

Biology, 07.10.2019 18:30

Social Studies, 07.10.2019 18:30

History, 07.10.2019 18:30