
Chemistry, 21.07.2019 15:00, Dmoney7784
Give the percent yield when 28.16 g of co2 are formed from the reaction of 4.000 moles of c8h18 with 4.000 moles of o2. 2 c8h18(l) + 25 o2(g) → 16 co2(g) + 18 h2o(g) molar mass co2 = 44.01 g/mol

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:00, levelebeasley1
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2

Chemistry, 22.06.2019 19:40, trodgers0202
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests. which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3

Chemistry, 22.06.2019 20:30, trevorhenyan51
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1
Do you know the correct answer?
Give the percent yield when 28.16 g of co2 are formed from the reaction of 4.000 moles of c8h18 with...
Questions in other subjects:





Biology, 04.09.2020 06:01

Social Studies, 04.09.2020 06:01

Health, 04.09.2020 06:01

Medicine, 04.09.2020 06:01

Mathematics, 04.09.2020 06:01


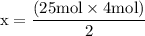 x = 50 moles of oxygen or O₂.
x = 50 moles of oxygen or O₂.  x = 112.64 gram of carbon dioxide
x = 112.64 gram of carbon dioxide Percentage yield = 25%
Percentage yield = 25%




