Chemistry, 21.07.2019 21:00, Anderson0300
P4(s) + 3o2(g) → p4o6(s) δh1 = -1,640.1 kj p4o10(s) → p4(s) + 5o2(g) δh2 = 2,940.1 kj what is the overall enthalpy of reaction for the equation shown below? round the answer to the nearest whole number. p4o6(s) + 2o2(g) -> p4o10(s)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:40, carebear60
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1

Chemistry, 22.06.2019 14:30, darkghostmist
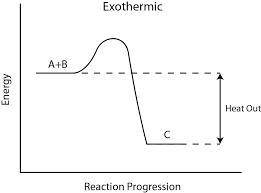
What type of reaction fuels the processes seen here?
Answers: 2


Chemistry, 22.06.2019 18:40, johnnysteeler9934
What is one real world example of a colligative property?
Answers: 2
Do you know the correct answer?
P4(s) + 3o2(g) → p4o6(s) δh1 = -1,640.1 kj p4o10(s) → p4(s) + 5o2(g) δh2 = 2,940.1 kj what is the ov...
Questions in other subjects:

Mathematics, 26.07.2019 23:30




Mathematics, 26.07.2019 23:30


Biology, 26.07.2019 23:30