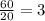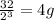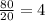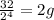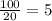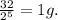The half-life of a radioactive element is defined as the time required by the specific isotope to decrease by half of its original value. The unobtanium after given time will be present as:
After 20 secs, 16 gramsAfter 40 secs, 8 gramsAfter 60 secs, 4 gramsAfter 80 secs, 2 gramsAfter 100 secs, 1 gram
Half-life is the time required by the unobtanium is the half of the reaction is completed. Half of the substance will be decomposed, such that:
Amount of substance left n half-lives = 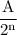
where A= initial amount of substance
Now,
n = number of half-lives = given time /half-life
Given,
1. Time = t = 20 seconds
Number of half-lives =
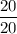
= 1Amount of the substance left after 1 half life =
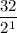
= 16 grams.
2.Time = t = 40 seconds
Number of half-lives =
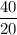
= 1Amount of the substance left after 1 half life =
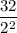
= 8 grams.
3.Time = t = 60 seconds
Number of half-lives =
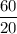
= 1Amount of the substance left after 1 half life =
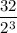
= 4 grams.
4.Time = t = 80 seconds
Number of half-lives =
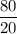
= 1Amount of the substance left after 1 half life =
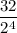
= 2 grams.
5.Time = t = 100 seconds
Number of half-lives =
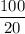
= 1Amount of the substance left after 1 half life =
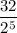
= 1 grams.
To know more about half-life, refer to the following link:
link
















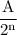
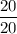 = 1Amount of the substance left after 1 half life =
= 1Amount of the substance left after 1 half life = 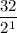 = 16 grams.
= 16 grams. 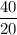 = 1Amount of the substance left after 1 half life =
= 1Amount of the substance left after 1 half life = 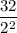 = 8 grams.
= 8 grams. 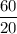 = 1Amount of the substance left after 1 half life =
= 1Amount of the substance left after 1 half life = 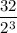 = 4 grams.
= 4 grams. 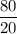 = 1Amount of the substance left after 1 half life =
= 1Amount of the substance left after 1 half life = 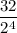 = 2 grams.
= 2 grams. 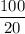 = 1Amount of the substance left after 1 half life =
= 1Amount of the substance left after 1 half life = 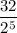 = 1 grams.
= 1 grams. 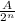
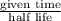
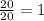
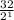 =16 g.
=16 g.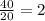
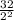 =8g.
=8g.