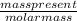Chemistry, 22.07.2019 20:00, brookeanne723
If you wanted to neutralize 4.00g naoh, what volume (l) of 1.0 m hcl would you need?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, darg3990rgp2t0r2
Does anyone know a lot about how to: - calculate mass of magnesium metal - calculate the actual yield of magnesium oxide - calculate the theoretical yield of mgo - calculate the percent yield of mgo - determine the percent yield of mgo - determine the average percent yield of mgo i had to do an online lab and its asking these questions but i have no idea where to start or how to be able to find these things. i can post the chart of the data from the lab or if you can tell me exactly how i can find each.
Answers: 3



Chemistry, 23.06.2019 06:30, kaitlynk0
Which of the following is true about the products formed during photosynthesis? (5 points) select one: a. they have the same mass as the mass of reactants. b. they are the same set of compounds as the reactants. c. they have more mass than the mass of reactants. d. they are chemically the same as the reactants.
Answers: 1
Do you know the correct answer?
If you wanted to neutralize 4.00g naoh, what volume (l) of 1.0 m hcl would you need?...
Questions in other subjects:

Mathematics, 06.08.2021 23:00


Mathematics, 06.08.2021 23:00





History, 06.08.2021 23:00