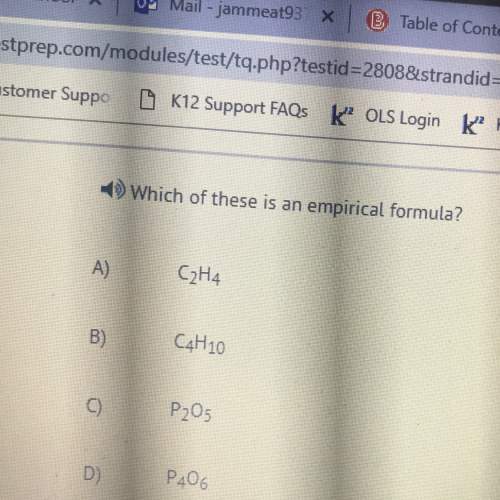
Chemistry, 23.07.2019 06:00, labrandonanderson00
During a temperature increase from -120.00 degrees celsius to 250.00 degrees celsius, a volume of of 2300 l is created. what was the original volume?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, angemango3423
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 11:00, familyvazquez7
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3

Chemistry, 22.06.2019 12:40, valenzueladomipay09u
How does concentration affect reaction rate
Answers: 2

Chemistry, 22.06.2019 13:50, kelonmazon2492
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
Do you know the correct answer?
During a temperature increase from -120.00 degrees celsius to 250.00 degrees celsius, a volume of of...
Questions in other subjects:

Mathematics, 04.02.2020 14:52


Mathematics, 04.02.2020 14:52

History, 04.02.2020 14:52