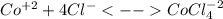In an aqueous chloride solution cobalt(ii) exists in equilibrium with the complex ion cocl42-. co2+(aq) is pink and cocl42-(aq) is blue. at low temperature the pink color predominates. at high temperature the blue color is strong. if we represent the equilibrium as: +(aq) + 4cl-(aq) cocl42-(aq) we can conclude that: 1. this reaction is: a. exothermic b. endothermic c. neutral d. more information is needed to answer this question.

Answers: 1
Similar questions

Chemistry, 11.10.2019 02:30, tucecoskun
Answers: 3

Chemistry, 31.10.2019 01:31, cambamm12
Answers: 1

Chemistry, 09.11.2019 04:31, jsph32
Answers: 2
Do you know the correct answer?
In an aqueous chloride solution cobalt(ii) exists in equilibrium with the complex ion cocl42-. co2+(...
Questions in other subjects:

English, 13.11.2019 15:31


Mathematics, 13.11.2019 15:31

Mathematics, 13.11.2019 15:31

English, 13.11.2019 15:31

History, 13.11.2019 15:31


Biology, 13.11.2019 15:31