Chemistry, 23.07.2019 21:00, janelisse199820
The combustion of gasoline produces carbon dioxide and water. assume gasoline to be pure octane (c8h18) and calculate the mass (in kg) of carbon dioxide that is added to the atmosphere per 1.3 kg of octane burned. (hint: begin by writing a balanced equation for the combustion reaction.)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, fastpitchhailey1354
An electrons position cannot be known precisely only it's probability of being in a certain location can be known
Answers: 1


Chemistry, 22.06.2019 17:30, tiffanyhmptn
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 22.06.2019 23:00, lilsnsbsbs
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2
Do you know the correct answer?
The combustion of gasoline produces carbon dioxide and water. assume gasoline to be pure octane (c8h...
Questions in other subjects:


English, 10.10.2019 08:00

English, 10.10.2019 08:00




Social Studies, 10.10.2019 08:00

Mathematics, 10.10.2019 08:00

Geography, 10.10.2019 08:00


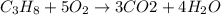
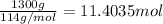
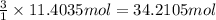 of carbon dioxide
of carbon dioxide