
Chemistry, 24.07.2019 13:00, gungamer720
Consider the following reaction: 2 h2s (g) two arrows stacked on top of each other. the top arrow points to the right. the bottom arrow points to the left. 2h2 (g) + s 2 (g) which answer represents the equilibrium constant for this reaction? the concentration of h subscript two to the exponent 2 multiplied by the concentration of s2 in the numerator all over the concentration of h2s to the exponent 2 in the denominator. the concentration of h subscript two multiplied by the concentration of s2 in the numerator all over the concentration of h subscript two s in the denominator. two times the concentration of h subscript two multiplied by the concentration of s subscript two in the numerator all over two times the concentration of h subscript two s in the denominator. the concentration of hsubscript two s to the exponent 2 in the numerator all over the concentration of h subscript two multiplied by the concentration of s subscript two in the denominator.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:00, uniqueray33
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

Chemistry, 22.06.2019 14:00, JJlover1892
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 15:00, raeprince9213
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1
Do you know the correct answer?
Consider the following reaction: 2 h2s (g) two arrows stacked on top of each other. the top arrow p...
Questions in other subjects:

Mathematics, 29.06.2019 00:00

Mathematics, 29.06.2019 00:00


History, 29.06.2019 00:00





History, 29.06.2019 00:00


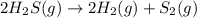
![K_c=\frac{[H_2]^2\times [S_2]}{[H_2S]^2}](/tpl/images/0127/3686/13e21.png)
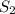 in the numerator all over the concentration of
in the numerator all over the concentration of 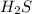 to the exponent 2 in the denominator.
to the exponent 2 in the denominator.



