50 points if right brainlest
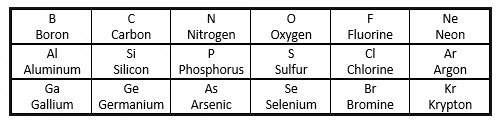
a section of the periodic table is shown below: which of t...


Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, brapmaster764
What is the formula that this ionic compounds could form sr2+p3-o2-
Answers: 3

Chemistry, 22.06.2019 01:00, angelteddy033
Which of the following is not a true statement about dwarf planets? a the kuiper belt contains comets, asteroids, and dwarf planets. b ceres is a dwarf planet located in the kuiper belt. c the largest known dwarf planet in the solar system is named eris.
Answers: 2

Chemistry, 22.06.2019 21:20, carlydays4403
The organs inside the body and how they function together
Answers: 3

Chemistry, 23.06.2019 16:00, emilystartk
Challenge question: this question is worth 6 points. as you saw in problem 9 we can have species bound to a central metal ion. these species are called ligands. in the past we have assumed all the d orbitals in some species are degenerate; however, they often are not. sometimes the ligands bound to a central metal cation can split the d orbitals. that is, some of the d orbitals will be at a lower energy state than others. ligands that have the ability to cause this splitting are called strong field ligands, cnâ’ is an example of these. if this splitting in the d orbitals is great enough electrons will fill low lying orbitals, pairing with other electrons in a given orbital, before filling higher energy orbitals. in question 7 we had fe2+, furthermore we found that there were a certain number (non-zero) of unpaired electrons. consider now fe(cn)6 4â’: here we also have fe2+, but in this case all the electrons are paired, yielding a diamagnetic species. how can you explain this?
Answers: 2
Do you know the correct answer?
Questions in other subjects:




Biology, 20.08.2020 20:01


History, 20.08.2020 20:01

Business, 20.08.2020 20:01

Arts, 20.08.2020 20:01


Mathematics, 20.08.2020 20:01