
Chemistry, 25.07.2019 06:30, kaydeemyylady
Achemistry lab needs to make 100 gallons of an 18% acid solution by mixing a 12% acid solution with a 20% solution. find the number of gallons needed of each solution.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, KieraKimball
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 00:30, xoxokaydavis5837
You have 125g of a certain seasoning and are told that it contains 76.0 g of salt what is the percentage of salt by mass in this seasoning
Answers: 1

Chemistry, 22.06.2019 17:30, nijanicole164
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2

Chemistry, 23.06.2019 03:30, memester74
Scientists often deal with numbers that are either very large or very small. for example, the radius of the sun is approximately 696,000 kilometers, while bacterial cells are as small as 1.9 × 10-4 millimeters. express each of these numbers in an alternate form.
Answers: 3
Do you know the correct answer?
Achemistry lab needs to make 100 gallons of an 18% acid solution by mixing a 12% acid solution with...
Questions in other subjects:

History, 04.10.2021 14:00



Mathematics, 04.10.2021 14:00


English, 04.10.2021 14:00


Mathematics, 04.10.2021 14:00



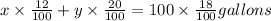 ...[2]
...[2]




