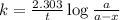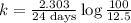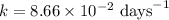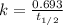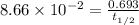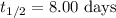Chemistry, 26.07.2019 03:00, lolomgwtfnvm4
5/3if 87.5 percent of a sample of pure 131 i decays in 24 days, what is the half-life of 131 i?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, Apple557
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1



Chemistry, 22.06.2019 19:30, simihehe
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
Do you know the correct answer?
5/3if 87.5 percent of a sample of pure 131 i decays in 24 days, what is the half-life of 131 i?...
Questions in other subjects:

English, 21.04.2020 00:14


Mathematics, 21.04.2020 00:14





Mathematics, 21.04.2020 00:14