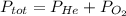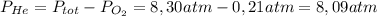He partial pressure of oxygen gas in our atmosphere is 0.21 atm. this is the partial pressure at which human lungs have evolved to be able to breathe this gas. a scuba diver, will thus still have to breath oxygen at this pressure even when diving way down in the water. if a mixture of helium and oxygen (heliox) in his tank is at a pressure of 8.30 atm, what must the partial pressure be of helium to keep the partial pressure of oxygen at 0.21 atm?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:00, graciewyatt6833
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 22.06.2019 19:30, liyahlanderson2232
Astring vibrates with a frequency of 10 hz. why can't a person hear the sound waves produced by the vibrating string, no matter how large the amplitude of the waves? out! this is homework and due tomorrow! you so much!
Answers: 2

Chemistry, 22.06.2019 21:30, djdjdjdbdbjx
What is another way to determine mass times acceleration?
Answers: 1
Do you know the correct answer?
He partial pressure of oxygen gas in our atmosphere is 0.21 atm. this is the partial pressure at whi...
Questions in other subjects:

History, 11.12.2021 03:10

Mathematics, 11.12.2021 03:10

Mathematics, 11.12.2021 03:10




Computers and Technology, 11.12.2021 03:10

Physics, 11.12.2021 03:10

Mathematics, 11.12.2021 03:10

Mathematics, 11.12.2021 03:10