
Chemistry, 27.07.2019 01:00, mamieengler
The equation below shows hydrogen reacting with oxygen to produce water. 2h2+o2> 2h2o if 16mol of oxygen were reacted with excess hydrogen gas, how many moles of water would be produced?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, shaylawaldo11
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3


Chemistry, 22.06.2019 11:40, Wemaybewrong
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1
Do you know the correct answer?
The equation below shows hydrogen reacting with oxygen to produce water. 2h2+o2> 2h2o if 16mol of...
Questions in other subjects:


History, 17.04.2021 23:40






Computers and Technology, 17.04.2021 23:40



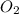 = 16 mole
= 16 mole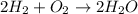
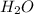
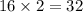 moles of
moles of 



