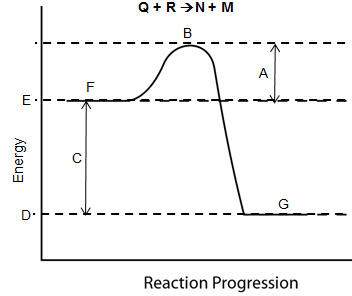
Consider the reaction pathway graph below.
which inference is supported by evidence in t...

Chemistry, 03.01.2020 00:31, jayybrain6337
Consider the reaction pathway graph below.
which inference is supported by evidence in the graph?
a and c show that the reaction enthalpy is less than the activation energy.
g and e show that this reaction is an endothermic process with heat release.
b and f show that the activated complex has less energy than the reactants.
e and d show that the energy of the products is less than that of the reactants.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3


Chemistry, 23.06.2019 00:40, joe7977
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
Do you know the correct answer?
Questions in other subjects:










Mathematics, 22.11.2019 22:31






