
Which characteristics of metal atoms explain why valence electrons in a metal are delocalized? metal atoms are large and have high electronegativities. metal atoms are small and have low electronegativities. metal atoms are large and have low electronegativities. metal atoms are small and have high electronegativities.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:50, Edwardwall
Elements in group 2 are all called alkaline earth metals. what is most similar about the alkaline earth metals?
Answers: 1

Chemistry, 22.06.2019 20:00, jalenevoyles
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 21:30, jpimentel2021
Under which circumstances are kp and kc equal for the reaction aa(g)+bb(g)⇌cc(g)+dd(g)?
Answers: 2

Chemistry, 23.06.2019 00:30, kylee65
What are the advantages of using the metric system? designed as a decimal system making conversions simpler more accurate system of measurement has prefixes that correspond to an amount to use with all base units used by the entire scientific community
Answers: 2
Do you know the correct answer?
Which characteristics of metal atoms explain why valence electrons in a metal are delocalized? met...
Questions in other subjects:

Biology, 14.11.2021 20:10


Mathematics, 14.11.2021 20:10

History, 14.11.2021 20:10




Mathematics, 14.11.2021 20:10


Biology, 14.11.2021 20:20


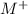 ions and group II elements are alkaline earth metals, they can give 2 electrons to form
ions and group II elements are alkaline earth metals, they can give 2 electrons to form 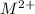 ions.
ions.



