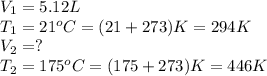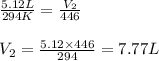Chemistry, 28.07.2019 07:00, bryson9604
Afixed quantity of gas at 21 °c exhibits a pressure of 752 torr and occupies a volume of 5.12 l. (a) calculate the volume the gas will occupy if the pressure is increased to 1.88 atm while the temperature is held constant. (b) calculate the volume the gas will occupy if the temperature is increased to 175 °c while the pressure is held constant.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, marissastewart533
City a and city b had two different temperatures on a particular day. on that day, four times the temperature of city a was 8â° c more than 3 times the temperature of city b. the temperature of city a minus twice the temperature of city b was â’3â° c. what was the temperature of city a and city b on that day? city a was 5â° c, and city b was 4â° c. city a was 3â° c, and city b was â’1â° c. city a was 8â° c, and city b was â’3â° c. city a was 5â° c, and city b was â’5â° c.
Answers: 2


Chemistry, 23.06.2019 04:31, 24swimdylanoh
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1
Do you know the correct answer?
Afixed quantity of gas at 21 °c exhibits a pressure of 752 torr and occupies a volume of 5.12 l. (a)...
Questions in other subjects:



English, 08.04.2020 19:05






Geography, 08.04.2020 19:05


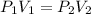
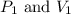 are initial pressure and volume.
are initial pressure and volume.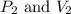 are final pressure and volume.
are final pressure and volume.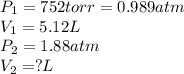
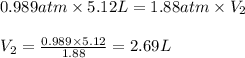
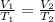
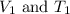 are the initial volume and temperature of the gas.
are the initial volume and temperature of the gas.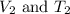 are the final volume and temperature of the gas.
are the final volume and temperature of the gas.