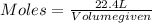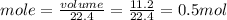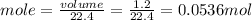Chemistry, 29.07.2019 14:00, brycehelmke60811
The molar volume of a gas at stp, in liters, is . you can use the molar volume to convert 2 mol of any gas to l. you can also use the molar volume to convert 11.2 l of any gas to mol. avogadro’s law tells you that 1.2 l of o2(g) and 1.2 l of no2(g) are of moles of gas.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, fireyking19
If the root word engage means “to connect with something,” what does the word disengage mean in the following sentence? he disengaged the gears by stepping on the clutch pedal. a.added more engine powerb. activated a connection to the pedalc. stalled the engined. released a connection to the pedal
Answers: 1

Chemistry, 22.06.2019 02:00, cbelew0001ouje4i
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1


Chemistry, 22.06.2019 13:30, xojade
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1
Do you know the correct answer?
The molar volume of a gas at stp, in liters, is . you can use the molar volume to convert 2 mol of...
Questions in other subjects:




Social Studies, 21.08.2019 21:30

Mathematics, 21.08.2019 21:30

Physics, 21.08.2019 21:30

History, 21.08.2019 21:30


Chemistry, 21.08.2019 21:30

English, 21.08.2019 21:30