
Chemistry, 29.07.2019 16:30, angelinacortes11
Consider the unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α, β, γ, δ = request answer part b determine how many moles of o2 are required to react completely with 6.4 moles c6h14. express your answer using two significant figures. n = mol

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:30, elijah1090
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 16:10, nauticatyson9
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1


Chemistry, 23.06.2019 02:40, towelmearowel
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3
Do you know the correct answer?
Consider the unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) par...
Questions in other subjects:



Geography, 27.02.2021 21:00

Social Studies, 27.02.2021 21:00

Arts, 27.02.2021 21:00

Mathematics, 27.02.2021 21:00

Mathematics, 27.02.2021 21:00

Chemistry, 27.02.2021 21:00

English, 27.02.2021 21:00

Mathematics, 27.02.2021 21:00


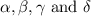 are 2, 19, 12 and 14 respectively.
are 2, 19, 12 and 14 respectively.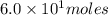 of
of 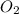 are required to react with 6.4 moles of
are required to react with 6.4 moles of 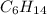 .
.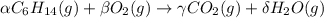
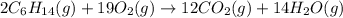
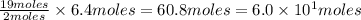 of
of 



