
Chemistry, 30.07.2019 18:00, rudondo4747
)calculate the molarity of a solution prepared by dissolving 117 g of sodium chloride (nacl) in enough water to make 2.7 liters of solution.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, penny3109
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 09:00, triddi666
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

Chemistry, 22.06.2019 09:30, andrejr0330jr
What is the molar mass of potassium nitrate, kno3
Answers: 1
Do you know the correct answer?
)calculate the molarity of a solution prepared by dissolving 117 g of sodium chloride (nacl) in enou...
Questions in other subjects:

Biology, 28.05.2021 09:50




Computers and Technology, 28.05.2021 09:50

Chemistry, 28.05.2021 09:50

Spanish, 28.05.2021 09:50


Health, 28.05.2021 09:50


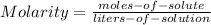 --- (A)
--- (A)



