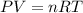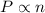Which of the following would double the pressure on a contained gas at constant temperature?
...

Chemistry, 26.08.2019 12:20, veronica25681
Which of the following would double the pressure on a contained gas at constant temperature?
a. doubling the volume of the container
b. halving the number of particles in the container
c. doubling the number of particles in the container
d. none of the above

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:00, amariyanumber1923
19. at high pressures, how does the volume of a real gas compare with the volume of an ideal gas under the same conditions, and why? eman- it is much less because real gas partides are not moving. there is no difference because the gas laws are always obeyed. it is much less because at high pressures the temperature drops. it is much greater because real gas partides take up space.
Answers: 1

Chemistry, 22.06.2019 04:30, KarenH3512
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1

Chemistry, 22.06.2019 07:20, rscvsdfsrysas1857
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 09:30, janetexcoelho
What does the mass of 0.7891 mol of ferric oxide (fe2o3)
Answers: 1
Do you know the correct answer?
Questions in other subjects:

History, 07.12.2020 15:30

Mathematics, 07.12.2020 15:30

History, 07.12.2020 15:30

Social Studies, 07.12.2020 15:30

Social Studies, 07.12.2020 15:30

History, 07.12.2020 15:30

Computers and Technology, 07.12.2020 15:30

Mathematics, 07.12.2020 15:30

Mathematics, 07.12.2020 15:30