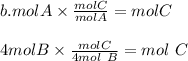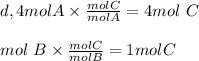Chemistry, 01.08.2019 21:40, milkshakegrande101
Look at the following hypothetical reaction: a + 4b - c + 3d. assume that the equation is balanced. which of the following mole ratios will produce the most product? a) a/b b) a/4b c) 3a/4b d) 4a/b

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 01:10, minasotpen1253
Volume is a measurement of how fast particles of a substance are moving
Answers: 3


Chemistry, 23.06.2019 08:30, vett072804
Benzonitrile (c6h5cn) is reduced to two different products depending on the reducing agent used. treatment with lithium aluminum hydride followed by water forms k, which has a molecular ion in its mass spectrum at 107 and the following ir absorptions: 3373, 3290, 3062, 2920, and 1600 cm-1. treatment with a milder reducing agent forms l, which has a molecular ion in its mass spectrum at 106 and the following ir absorptions: 3086, 2850, 2820, 2736, 1703, and 1600 cm-1. l shows fragments in its mass spectrum at m/z = 105 and 77. propose structures for k and l and choose an explanation for how this could be concluded.
Answers: 3

Chemistry, 23.06.2019 12:30, lindseylewis313
When utilizing a transmission electron microscope, why is it necessary to stain the specimen with heavy metal salts?
Answers: 1
Do you know the correct answer?
Look at the following hypothetical reaction: a + 4b - c + 3d. assume that the equation is balanced....
Questions in other subjects:

English, 01.08.2021 09:00

Mathematics, 01.08.2021 09:00



Mathematics, 01.08.2021 09:00

Mathematics, 01.08.2021 09:00


Mathematics, 01.08.2021 09:00

Biology, 01.08.2021 09:00


 .
.